
Three experiments led him to this. The model was named after a common english dessert which has raisins dispersed throughout a matrix of cake. He advanced the idea that cathode rays are really streams of very small pieces of atoms. This led him to hypothesise the plum pudding model of the atom, where a sea of negative particles (electrons, as we now call them) were surrounded by a cloud of positive charge to balance them out. During the experiment, he discovered the electron, which is one of the. He theorised that to balance out this negative charge present in these small particles, they must be surrounded by a positively charged matter that made up the matrix of the atom. One of the phenomenal contributions of Thomson to science is his cathode ray experiment. Cathode rays (blue) emitted by the cathode on the left were defelcted by an electric field (yellow) in the center. He further concluded that these small particles were building blocks of larger particles. English: Diagram of JJ Thomsons experiment with cathode rays. This led him to conclude that these particles were a smaller piece of matter than the atom itself. He measured the mass of the particles and discovered they were 1800 times smaller than that of the element hydrogen. of the kinematics of the particles, deflected by a field as explained here. When you are ready to start the experiment, click on the. Your job is to measure the deflection of the beam under different conditions and then determine the ratio of the charge of the particles in the beam to the mass of the particles in the beam. Thomson concluded that these rays were not light but instead made of negatively charged particles. Thomsons experiments on cathode rays were important for the determination. In this lab we will be looking at a model of the Thomson experiment using a Cathode Ray Tube. 
When the cathode ray was attracted toward the positively charged plate, he realized that the rays must be made up of negatively charged particles. Thomson decided to set up additional electric plates around the cathode ray to determine whether the cathode beams were charged. Chemists called this particle flow “cathode rays,” and they were able to detect them by placing a material called phosphors in the tube. Most of the air was removed from the tube, but when the voltage is applied, the remaining particles flowed from the cathode to the anode. To create a cathode ray tube, Thomson applied a voltage to one side of a sealed glass container with two electrodes. Thomson discovered that there was a particle smaller than an atom - the electron - through his work with cathode ray tubes. However, in 1897, the English physicist J. The work of Avogadro on the volumes of gases and Robert Brown on Brownian motion further supported this hypothesis. This theory was established by Dalton and the atomists who stated that matter was made up of small indivisible particles known as elements which varied in their nature. Thomson, Cathode rays (Friday evening meeting of the. 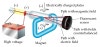
The plum pudding model was the first model to suggest that atoms were not the smallest unit of matter, and that they may be made up of both positive and negatively charged subatomic particles.įor a long time, atoms were thought to be the smallest unit of matter. his corpuscle hypothesis, and in section 4 I discuss the experiments of 1897. What did the plum pudding model suggest about atoms? Instead of a sea of positive charges surrounding negative charged particles, atoms are actually organized with positive and neutral particles at their core (the nucleus) surrounded by orbitals of electrons.ģ. While the plum pudding model was the first to suggest that atoms are made up of charged particles, the plum pudding model is not entirely correct. This work was later expanded upon by chemists throughout the 19th century and beyond to create the modern model of the atom, which arose after the rise and fall of many partial theories.ġ. The modern atomic model started to take shape with the work of Lavoisier and Dalton who formulated the concept of elements as unique iterations of atoms which can be combined in multiple ways to create a variety of substances. \): Millikan’s experiment measured the charge of individual oil drops.Refresher: The atomic model originated in the 5th century BC when Greek philosophers hypothesized that all matter was composed of indivisible particles.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
